- Why Clean Air Matters in Pharma Manufacturing
- Where Compressed Air Is Used in Pharma
- Air Quality Standards and ISO 8573-1 Explained
- Ambient Air and Leak Detection Instruments
- Humidity and Oil Moisture Sensors
- Flow Meters for Compressed Air
- Wet Air Flow Sensors
- Dew Point Monitoring for Compliance
- Particle Counters and Oil Check Monitoring Systems
- Oil-Free Air Compressors: Choosing the Right Equipment for Your Pharmaceutical Cleanroom
- Oil-Free vs. Oil-Lubricated Compressors with Filtration
- Maintain Compliance with Continuous Monitoring
Why Clean Air Matters in Pharma Manufacturing
Clean, contaminant-free air is essential in pharmaceutical production. Every air compressor in pharmaceutical industry operations must deliver consistent pressure and purity to maintain product integrity and meet regulatory expectations.
Compressed air in pharmaceutical facilities is used in two ways:
- Direct contact applications, such as capsule filling, coating, or packaging, where air may come into contact with the product.
- Indirect contact applications, such as air used to power pneumatic systems or control cleanroom processes.
Maintaining clean compressed air helps prevent contamination and equipment downtime. Although the FDA has not established specific clean air limits, facilities are expected to meet ISO 8573-1 purity classes and cGMP guidelines. Compressed Air Systems (CAS) supplies complete pharmaceutical compressed air solutions, including oil-free and oil-lubricated compressors, dryers, filters, and monitoring instruments that support pharmaceutical manufacturing environments.

WHERE COMPRESSED AIR IS USED IN PHARMA
Compressed air is indispensable across nearly every stage of pharmaceutical manufacturing. Typical applications include:
AIR QUALITY STANDARDS AND ISO 8573-1 EXPLAINED
The ISO 8573-1 standard defines the maximum permissible levels of solid particles, water, and oil in compressed air. Pharmaceutical manufacturers typically require Class 0, 1, or 2 purity depending on whether air comes into direct contact with products.
| ISO Class | Solid Particles (µm) | Water (Pressure Dew Point) | Oil (mg/m³) | Common Pharma Applications |
|---|---|---|---|---|
| Class 0 | As specified by user | As specified by user | As specified | Sterile manufacturing, injectables |
| Class 1 | ≤ 0.1 | ≤ −70 °C (−94 °F) | ≤ 0.01 | Tablet compression, filling, packaging |
| Class 2 | ≤ 1.0 | ≤ −40 °C (−40 °F) | ≤ 0.1 | Lotion manufacturing, indirect use |
| Class 3 | ≤ 5.0 | ≤ −20 °C (−4 °F) | ≤ 1 | Conveyors, pneumatic tools (non-contact) |
| Class 3–4 | ≤ 15.0 | ≤ +3 °C (37.4 °F) | ≤ 5 | Utility air (non-cleanroom areas) |
Maintaining these levels typically involves a combination of dryers and multi-stage filtration. CAS supplies air compressors and purification systems that meet the compressed air quality standards that pharmaceutical facilities require.
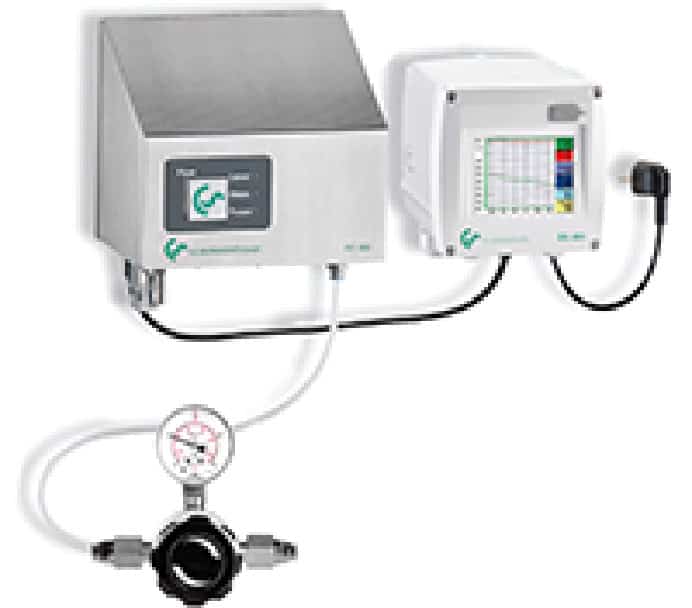
OIL-FREE AIR COMPRESSORS: CHOOSING THE RIGHT EQUIPMENT FOR YOUR PHARMACEUTICAL CLEANROOM
In pharmaceutical cleanrooms, even trace oil contamination can jeopardize sterile products and regulatory compliance. For this reason, oil-free air compressors are the preferred choice in critical environments where air purity is paramount.
Oil-free systems eliminate the risk of internal oil carryover, helping manufacturers maintain ISO Class 0 air quality and meet FDA cGMP and FSMA standards. They are ideal for processes involving direct product contact, such as tablet coating, capsule filling, and aseptic packaging.
When selecting compressors for pharmaceutical use, consider:
OIL-FREE VS. OIL-LUBRICATED COMPRESSORS WITH FILTRATION
Oil-free compressors eliminate the risk of oil contamination, while oil-lubricated compressors with advanced filtration offer comparable purity at lower cost.
| Feature | Oil-Free Compressor | Oil-Lubricated + Filtration |
|---|---|---|
| Initial Cost | High (up to 2x higher) | Lower |
| Maintenance Costs | High (sensitive components, costly repairs) | Moderate (routine oil & filter changes) |
| Compressor Lifespan | Shorter (due to more friction) | Longer (with regular service) |
| Compressed Air Purity | Class 0 possible (still requires intake filtration) | Class 0–2 possible with advanced filtration |
| Risk of Oil Carryover | Minimal (no internal oil) | Low (with proper coalescing/carbon filters) |
| Energy Efficiency | Lower (due to greater rotor friction) | Higher (more efficient sealing) |
| Best For | Ultra-critical sterile environments (injectable drugs, cleanrooms) | Most pharmaceutical manufacturing environments |
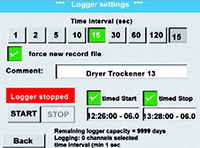
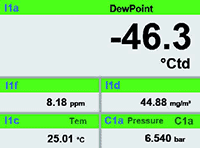
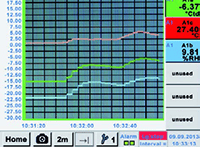
MAINTAIN COMPLIANCE WITH CONTINUOUS MONITORING
Continuous, sensor-based monitoring simplifies air quality validation and supports FDA and ISO documentation requirements. By integrating leak, humidity, oil moisture, wet air flow, and other monitoring tools with air compressors, pharmaceutical manufacturers can ensure every system meets the necessary air quality standards.
Compressed Air Systems supplies the compressors, filtration, and monitoring equipment needed to maintain compliant, efficient, and high-purity air throughout pharmaceutical production.
Contact our team today to discuss your air requirements.